A case report of haploidentical hematopoietic stem cell transplantation for diffuse large B-cell lymphoma developing in the course of immunosuppressive therapy after kidney transplantation
Elena N. Parovichnikova, Vera A. Vasilyeva, Olga A. Aleshina, Mariya V. Dovydenko, Larisa A. Kuzmina, Mikhail Yu. Drokov, Irina A. Lukyanova, Vera V. Troitskaya, Evgenii E. Zvonkov, Alla M. Kovrigina, Valery G. Savchenko
National Medical Research Center for Hematology, Moscow, Russia
Correspondence:
Dr. Vera A. Vasilyeva, PhD, MD, Head Immunochemotherapy Department for BMT patients, National Medical Research Center for Hematology, 4 Novyi Zykovskii Lane, Moscow, 125167, Russia
Phone: +7 (916) 262-23-16
E-mail: vasilievaVA4@mail.ru
Citation: Parovichnikova EN, Vasilyeva VA, Aleshina OA et al. A case report of haploidentical hematopoietic stem cell transplantation for diffuse large B-cell lymphoma developing in the course of immunosuppressive therapy after kidney transplantation. Cell Ther Transplant 2022; 11(1): 50-57.
Accepted 18 March 2022
Summary
Post-transplant lymphoproliferative disease (PTLD) is one of the most common complications after organ transplantation. The main goal of PTLD treatment is to achieve stable remission of the disease and preserve the transplanted organ. However, it is not uncommon that after PTLD therapy immunosuppressive therapy for the transplanted organ is returned, which may again lead to a recurrence of PTLD. Autologous hematopoietic stem cell transplantation is a rare option for treatment PTLD. The role of allogeneic hematopoietic stem cell transplantation (HSCT) in PTLD therapy after solid organ transplantation has not been determined, due to the high risks of toxicity for the transplanted organ, the development of a graft-versus-host disease may lead to rejection of the transplanted solid organ, and the presence of a solid organ transplant may increase the rejection of hematopoietic stem cells.
We have described a case report of haploidentical hematopoietic allogeneic stem cell transplantation with TCR αβ+/CD19+ cell depletion for a patient with second remission of diffuse large B-cell lymphoma associated with immunosuppressive therapy for a previously performed allogeneic cadaveric kidney transplantation.
Keywords
Post-transplant lymphoproliferative disease, hematopoietic allogeneic stem cell transplantation, haploidentical, TCR αβ+/CD19+ cell depletion.
Introduction
The development of solid organ transplantation and the subsequent lifelong immunosuppressive therapy (IST) is naturally accompanied by the development of complications associated with a specific spectrum of infections, organ dysfunctions, and also with the occurrence of tumor diseases of various etiology. Increasing the number of patients who underwent transplantation of kidneys, liver, heart, lungs, etc. results in the number of patients with post-transplant malignant neoplasms [1]. The most common of them are neoplastic diseases of the hemopoietic system and, mainly, various malignant lymphoproliferative diseases. Even separate forms of these types of tumors which is associated with previous long-term immunosuppression are distinguished in the modern WHO classification [2].
The risk of developing post-transplant lymphoproliferative disease (PTLD) increases by 20-120% in patients after solid organ transplantation compared with the general population and depends on the intensity of IST. PTLD was first described in 1968 by Doak P. B. in two patients after cadaveric kidney transplantation and IST with azathioprine and prednisolone [3]. The term PTLD was introduced by T. E. Starzl in 1984 [4]. The role of the Epstein-Barr virus (EBV) in the pathogenesis of PTLD was described in 1981 [5]. The developing of this disease most frequently associated with solid organ transplantation like heart, lung, intestine and multi-organ transplantation, while this complication develops less frequently in kidney and liver recipients [6, 7]. There was no increase in the incidence of PTLD when cyclosporine was used in comparison with therapy with azathioprine in combination with glucocorticosteroids. Tacrolimus also increases the risk of this complication [7]. Immunosuppressive therapy with antithymocyte globulin (ATG) increases the risk of PTLD within the first year after solid organ transplantation, and no such dependence was found when antibodies to the IL-2 receptor were used [7]. It has been established that in the pathogenesis of PTLD, the presence of certain human leukocyte antigens (HLA) in a patient is important. If alleles HLA-A26 and HLA-B38 are detected in a recipient, the risk of PTLD development increases, while donor haplotypes HLA-A1, HLA-B8, and HLA-DR3 were identified as factors in which this complication develops less frequently [8]. Other studies have identified an increased risk of PTLD in a recipient with HLA-A2, HLA-A11, HLA-B5, HLA-B18, HLA-B21, and HLA-B35 [9, 10].
The clinical manifestations of PTLD are often nonspecific. Involvement of the gastrointestinal (GI) tract, lungs, skin, bone marrow, and central nervous system (CNS) is common [11-15]. CNS involvement is present in about 10-15% of all PTLD cases [11]. B-symptoms such as pyrexia, sweating and weight loss are also common. Damage to the transplanted organ is noted in about a third of all PTLD cases [13]. According to the WHO classification 2017, there are the following types of PTLD: early lesions (plasmacytic hyperplasia, infectious mononucleosis, and florid follicular hyperplasia) – 5% of all PTLD, polymorphic PTLD – 15-20%, monomorphic B-cell neoplasm (diffuse large B-cell lymphoma, Burkitt lymphoma, plasma cell myeloma and other) – more than 70% of all PTLD, monomorphic T/NK-cell neoplasms PTLD (peripheral T-cell lymphoma and other) – less than 5%, and classic Hodgkin lymphoma PTLD – less than 5% [2].
The main goal of PTLD treatment is to achieve stable remission of the disease and preserve the transplanted organ. Due to the clinical and pathological heterogeneity of PTLD (type of transplant, risk of rejection and concomitant diseases, tumor burden, etc.), there is no standardized approach to treatment. Nevertheless, the key approach is the reduction of IST: complete cancellation or partial (by 50-75%) reduction the dose of immunosuppressive drugs to prevent rejection of the transplanted organ. In this case, the response is achieved in about half of the patients, but long-term remissions are rare [12, 14-16]. Other methods that can be used in the treatment of PTLD are monoclonal antibodies to the CD20 antigen (rituximab) as monotherapy or in combination with chemotherapy, surgery, and radiation therapy. In the event that the development of PTLD is associated with EBV infection, antiviral therapy (intravenous immunoglobulins) and/or EBV-specific T-lymphocytes are used, autologous or allogeneic hematopoietic stem cells transplantation are used less often as a consolidating treatment [11, 13, 14, 16.17]. Overall survival of patients after PTLD therapy has significantly improved in recent years, by the using of monoclonal antibodies. Mortality rate earlier was reached 50-70%, nowadays the 3-year overall survival rate is 60-70%, according published studies in recent years [7, 13, 14].
The role of allogeneic hematopoietic stem cells (allo-HSCT) transplantation in PTLD therapy after solid organ transplantation has not been determined. The use of high-dose chemotherapy can be accompanied by high toxicity for the transplanted organ. Graft-versus-host disease (GVHD), which develops after allo-HSCT, can contribute to the rejection of a transplanted solid organ. And the presence of a solid organ transplant before allo-HSCT may increase the probability of rejection of hematopoietic stem cells. Only a few successful clinical cases of allo-HSCT in PTLD after solid organ transplantation have been described [1, 18-20].
The aim of this work: to present a clinical case of a patient in whom transplantation of an allogeneic cadaveric kidney was complicated by the development of PTLD, which was initially successfully treated, but due to the ongoing IST, a relapse was established, which required HSCT from a haploidentical donor. Clinical case is presented with the consent of the patient.
Clinical case description
Patient B., female, the disease manifested at the age of 9 years, in 1996 with proteinuria (3 g/day) and increased arterial pressure up to 160/90 mm Hg. Acute glomerulonephritis was diagnosed, and prednisolone therapy was started. In 1998, the diagnosis of mesangioproliferative glomerulonephritis was confirmed by histological examination of a kidney biopsy. Progression to the terminal stage of chronic renal failure (stage 5) was documented in 2003, hemodialysis was started, with arteriovenous fistula arranged. Transplantation of allogeneic cadaveric kidney was performed at the N. V. Sklifosovsky Research Institute for Emergency Medicine in April 2011. Delayed recovery of renal transplant function was noted; hemodialysis was carried out within a month after the organ transplantation. Tacrolimus, mycophenolate mofetil, methylprednisolone were applied as immunosuppressive therapy. Blood creatinine level in the post-transplant period was 110-130 μmol/L, glomerular filtration rate (GFR), according to the CKD-EPI formula, was 48-61 ml/min/1.73 m2. Due to the pregnancy planning, mycophenolate mofetil was replaced by azathioprine in 2016. Pancytopenia was detected in June 2017, but the dose of immunosuppressive drugs was not reduced. Since the end of July 2017, the patient had febrile body temperature, decreasing body weight, dry cough, increased abdominal volume, and irregularity in the menstrual cycle, but the patient did not seek medical asssistance.
In August 2017 the examination revealed proteinuria 2.3 g/l, hypoalbuminemia 28 g/l, high level of blood creatinine (214 μmol/l) and C-reactive protein (115 mg/dl), an increased LDH activity to 923 U/l, ESR up to 70 mm/h, an increased IgM content in the blood (1288 mg/dl), mainly, due to IgMλ. Pancytopenia still persisted (leukocytes 1.9×109/l, platelets 90×109/l). Antibiotic therapy was carried out for suspected infection, azathioprine was canceled. Hepatosplenomegaly was revealed on the CT examination of abdominal organs. Given these findings, lymphoproliferative disease was suggested, thus requiring special examination.
The patient was examined at National Research Center for Hematology at 23.08.2017. Infiltrative-ulcerative defects of the gastric mucosa were found during esophagogastroduodenoscopy. The data of histological and immunohistochemical studies of gastric biopsy revealed the substrate of extranodal diffuse large B-cell lymphoma (tumor cells monomorphically expressed CD 20). In addition, the data of histological and immunohistochemical studies of the bone marrow also was determined: interstitial-small-focal B-cell infiltration with the presence of clusters of large CD 20+ cells, intravascular foci of small CD 20+ B-cells. The expression of EBER (EBV-encoded RNA) was not detected, but the virus replication was detected in blood. Paraprotein M lambda, 21.3 g/l; Bens-Jones protein lambda 0.35 g/l was revealed in blood serum by immunochemistry. Hyperpdiloid karyotype and complex cytogenetic rearrangements involving chromosomes 3, 7, 4, 9, 10, 14, 15, 17, 18 were registered by standard cytogenetic study of the bone marrow samples. According to CT scan, hepatosplenomegaly was determined: the vertical size of the liver was 23.5 cm, the size of the spleen was 160×60×235 mm. Based on the obtained data, the diagnosis of PTLD was specified as a diffuse large B-cell lymphoma (PTLD-DLBCL) associated with immunosuppressive therapy, with involvement of liver, spleen, stomach and bone marrow.
Despite the presence of a transplanted organ, taking into the variant of the disease and the generally accepted practice of managing such patients, the IST was reduced: methylprednisolone and tacrolimus were completely canceled since 24.08.2017.
From 26.08.2017 to 30.08.2017, a pre-phase with dexamethasone and cyclophosphamide was carried out and a positive trend was noted as decreasing size of the liver and spleen. From 31.08.2017 to 05.09.2017, the 1st course of CHOD protocol in combination with rituximab (600 mg) was administered (total drug doses: cyclophosphamide 1200 mg, doxorubicin 75 mg, vincristine 2 mg, dexamethasone 80 mg). The treatment was complicated by febrile fever, oropharyngeal candidiasis, bilateral pleuropneumonia; the patient underwent massive antimicrobial therapy: imipenem/cilastatin, sodium colistimethate, fluconazole, acyclovir and intravenous immunoglobulin. In addition, the patient developed hypotension (up to 80/50 mm Hg). Subsequent examination revealed adrenal insufficiency, which required administration of hydrocortisone (2.5-5 mg per day). A partial remission of the disease was obtained after first R-CHOD. From 26.09.2017 to 11.01.2018, five R-CHOD courses were performed. A complete remission of PTLD-DLBCL was achieved after second course of R-CHOD as confirmed by PET-CT (Fig. 1).

Figure 1. CT dynamics of internal organs. A, in the debut of PTLD-DLBCL (August 2017); B, first remission of PTLD-DLBCL (January 2018); C, relapse of PTLD-DLBCL (January 2019); D, before haplo-HSCT (May 2019)
Due to the risk of renal transplant rejection, the patient resumed immunosuppressive therapy with tacrolimus (2 mg/day) and prednisolone (5 mg/day) after completing chemotherapy (6 courses), since April 2018. In December 2018, 10 months after the completion of therapy for PTLD-DLBCL, the patient had a febrile episode of up to 40°C. Ultrasound examination revealed a focal lesion in the liver. At that moment, no other signs of PTLD progression (based on bone marrow and gastric biopsies) were present. However, a month later, in January 2019, the febrile fever was again observed. The patient was admitted to the hospital in an extremely severe condition with signs of septic shock, bilateral pneumonia with respiratory failure, and evolving acute renal injury (blood creatinine levels reached 522 μmol/l). At the intensive care unit, the patient underwent massive antimicrobial therapy (meropenem, linezolid, sodium colistimethate, fluconazole, acyclovir), and hemodiafiltration were performed, with immunosuppressive therapy still continued. Additional examination revealed an increasing size of the liver (vertical size 24 cm) and spleen (121×54×175 mm) (Fig. 1). B-cell clonality was revealed in the gastric tissue biopsy, infiltration with tumor CD20+ cells was noted in the bone marrow biopsy, monoclonal secretion of M lambda in serum was determined in blood (5.3 g/l), a trace amount of the Bence-Jones protein lambda was detected in the urine, standard cytogenetic study of bone marrow and gastric tissue biopsies no mitosis was detected. Thus, after 11 months of achievement of PET-CT-negative remission and 9 months after the resumption of the IST, a relapse of PTLD-DLBCL was documented.
The severity of the patient's condition was due to both septic condition and emerging relapse of the disease, including specific liver damage. As a result, severe coagulation disorders developed, with re-appearing signs of adrenal insufficiency (decreased concentration of cortisol in blood serum: 90.47 nmol/L (reference range of 185-624 nmol/L), which required administration of hydrocortisone (2.5-5 mg per day), blood transfusions and management of blood clotting problems.
Due to PTLD-DLBCL relapse and severe infectious complications, despite presence of a transplanted organ, the IST was canceled 01.05.2019, and the antitumor effect was not achieved again. The patient's condition remained extremely serious, and according to vital indications, from 01.11.2019 to 15.01.2019 the pre-phase therapy with cyclophosphamide and dexamethasone was started. Positive clinical dynamics was noted, i.e., normalization of body temperature and decrease in the liver and spleen size few days after pre-phase therapy.
Thereafter, a modified course of chemotherapy with cytarabine (=4 g) + etoposide (∑ = 400 mg) combined with lenalidomide (∑ = 30 mg) was carried out from 16.01.2019 to 17.01.2019. Lenalidomide was included in the treatment, according to the previously published data on improvement of overall and progression-free survival when using it in the patients with DLBCL [21, 22]. At the control examination (08.02.2019) we have revealed a decreased size of the liver (vertical size 18 cm) and spleen (115×47×130 mm), complete regression of focal liver lesion, regression of hystological changes in the bone marrow biopsy, and only trace secretion of paraprotein IgM.
Two more courses using cytarabine + etoposide + lenalidomide were performed from 15.02.2019 to 06.05.2019. The second PET-CT-negative remission of PTLD-DLBCL was achieved (Fig. 1).
Thus, the young patient re-achieved PET-СТ negative remission of PTLD-DLBCL, and still required resumption of IST to prevent kidney transplant rejection, which would entail another relapse of the disease. Program therapy for patients in the second remission of the DLBCL implies to consolidate remission by autologous hematopoietic stem cell transplantation. However, our patient would have to return again immunosuppressive therapy to prevent kidney transplant rejection. Therefore, we decided to perform allo-HSCT to induce tolerance of the donor-derived immune cells to the transplanted kidney. The patient didn’t have a fully matched related or unrelated donor, since the HSCT was planned from a related haploidentical (haplo-HSCT) donor (mother), however, with TCRαβ +/CD19+ depletion of the graft. The choice of such a transplant approach was determined by the fact that it does not use long-term (minimum 6 months) immunosuppressive therapy. The last injection of IST drugs (tocilizumab and abatacept) was administered on day +30 after haplo-HSCT with TCRαβ+/CD19+ depletion. Following HLA typing of the patient and her mother was performed, HLA-B18 was identified in the HLA genotype of the patient. This allele is associated with increased incidence of PTLD. The donor was diagnosed with microcytic hypochromic anemia (Hb 101 g/L), she was observed for a long time at a hematologist with a diagnosis of iron deficiency anemia and was treated with iron preparations.
Pre-transplant conditioning regimen included: treosulfan (∑ = 45 g) + melphalan (∑ = 100 mg) + fludarabine (∑ = 225 mg). Haplo-HSCT was performed 14.05.2019 the number of CD34 + cells in the graft was 10.8×106/kg, the number of TCRαβ + lymphocytes was 271.5×103/kg. GVHD prophylaxis consisted of rituximab (∑ = 170 mg) + bortezomib (∑ = 4.2 mg) + tocilizumab (∑ = 460 g) + abatacept (∑ = 1740 mg), the patient did not receive any other immunosuppressive therapy. The early post-transplant period was complicated by febrile fever, mucositis, and neutropenic enterocolitis, positive effect was obtained on carbapenem therapy. The recovery of leukocytes was on day +13 after haplo-HSCT (3.0×109/l).
The patient was discharged from the hospital on day + 21 after haplo-HSCT. There were no problems with the functioning of the renal graft, during conditioning regimen and in the early post-transplant period too.
Early control examinations after haplo-HSCT (+1, +2 months) has shown bone marrow remission of the disease with normal function of erythropoiesis and 100% donor chimerism. However, despite the absence of ABO incompatibility between the patient and the donor (the patient and the donor have the same blood group: 0 (I) Rh + CcDee with differences in the MN system), 95% of erythrocytes had patient’s phenotype. Meanwhile, severe anemia persisted for 6 months after haplo-HSCT, requiring monthly blood transfusions. Therefore, additional tests were performed to search for possible causes of anemia (Table 1). Initially, there were signs of either iron deficient anemia, or anemia of chronic disease (25.06.2019). Parvovirus B19-associated anemia was excluded. However, in September 2019, the signs of microcytic hypochromic anemia worsened, there were a decrease in TIBC, LIBC and transferrin, along with increased ferritin level and normal serum iron content. Mild iron overload was determined by MRI. The clinical diagnosis was considered as follows: anemia of chronic conditions in a patient with transplanted kidney and multiple complications associated with therapy of the disease.
Table 1. Post-transplant monitoring of anemia
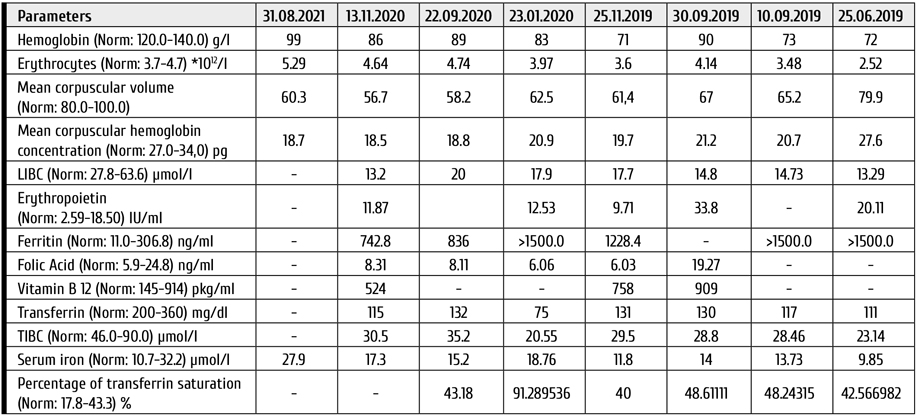

Figure 2. A: Slight increase in the light and heavy fractions of erythrocytes (abscissa, relative cell density, g/cm3). The average density of red blood cells is within normal limits. (B, C): Osmotic fragility (OFT) is altered before and after incubation (abscissa, % of NaCl in saline for RBC incubation)
Given the fact that before haplo-HSCT patient didn’t have any signs of microcytic hypochromic anemia, we have performed hemoglobin electrophoresis, density distribution of erythrocytes, and the osmotic fragility test (OFT) (Fig. 2). The content of the fraction of normal hemoglobin A was 95.9% and hemoglobin A2 was 4.1%.
According to these studies, clinical thalassemia was suspected, and during DNA diagnostic test was found heterozygous carriage of beta (+) – thalassemic mutation IVS1 + 5 G> T. A genetic study of an archived sample of the patient's bone marrow before haplo-HSCT was carried out: thalassemic mutation was not detectable. Thus, the patient was diagnosed with a heterozygous form of beta-thalassemia, which was a consequence of the reconstitution of donor hematopoiesis.
The examination of the haploidentical related donor, the mother, was carried out, and again, as before donation, moderate microcytic hypochromic anemia was revealed. A genetic study was performed, and it was confirmed that the mother is also a heterozygous carrier of the beta (+) – thalassemic mutation IVS1 + 5 G> T.
At the present time, 2.5 years have passed after haplo-HSCT, and 10.5 years after transplantation of allogeneic cadaveric kidney. In this case, the patient had a short course of immunosuppressive drugs (rituximab, bortezomib, tocilizumab, abatacept) only at the early post-transplant period, ceasing the IST at later terms. Immunological tolerance has been achieved: the patient has no signs of GVHD, and the kidney transplant is functioning normally. The patient has PET-CT-negative remission of PTLD-DLBCL. Despite the fact that the patient is a heterozygous carrier of the beta-thalassemia mutation, her primary disease was complicated by the anemia of chronic conditions, which required hemotransfusions for a year after haplo-HSCT. Currently, the patient is transfusion-independent.
Conclusion
Post-transplant lymphoproliferative disease is not such a rare complication after solid organ transplantation, since the immunosuppressive therapy performed increases the risks of developing lymphoid tumors.
We have described for the first time a case of haploidentical HSCT with TCRαβ+/CD19+ graft depletion in a patient with second remission of diffuse large B-cell lymphoma associated with immunosuppressive therapy for a transplant of allogeneic cadaveric kidney. In the literature, there are only a few descriptions of allo-HSCT from related or unrelated donors in PTLD after solid organ transplantation, and haploidentical HSCT with TCRαβ+/CD19+ graft depletion has not been previously described. This transplant mode was chosen due to absence of HLA-identical donors, second remission of PTLD-DLBCL, and the fact that the previous relapse was associated with resumption of immunosuppressive therapy (IST) for the transplanted kidney. Haploidentical HSCT with TCRαβ+/CD19+ depletion was performed, since the duration of immunosuppressive therapy is only one month after such procedure. The therapy we have chosen in relapsed PTLD-DLBCL allowed us to achieve a second complete PET-CT-negative remission, then was consolidation with haplo-HSCT, which led to development of immunological tolerance of the donor immune system to the previously transplanted cadaveric kidney and withdrawal of IST, which seemed to be the key factor of PTLD. Both kidney transplant and a bone marrow graft show adequate functioning without additional IST.
Another feature of this patient is that the donor for HSCT is a heterozygous carrier of beta-thalassemia mutation IVS1 + 5 G> T, which was not detected during the initial examination, manifesting with serum iron deficiency. There is no doubt that initial detection of beta-thalassemia trait in the donor could influence the donor choice, since the presence of this mutation and the transplanted kidney during the first year complicated the patient's quality of life after haplo-HSCT.
Conflict of interest
None reported.
References
- Doney KC, Mielcarek M, Stewart FM, Frederick R. Hematopoietic cell transplantation after solid organ transplantation. Biol Blood Marrow Transplant. 2015;21(12):2123-2128. doi: 10.1016/j.bbmt.2015.08.004
- Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J. WHO Classification of tumours of haematopoietic and lymphoid tissues – IARC [Internet]. Revised 4th edition. Lyon: IARC Press. Lyon: IARC Press; 2017.
- Doak PB, Montgomerie JZ, North JDK, Smith F. Reticulum cell sarcoma after renal homotransplantation and azathioprine and prednisone therapy. Br Med J. 1968; 4(5633): 746-748. doi: 10.1136/bmj.4.5633.746
- Starzl TE, Porter KA, Iwatsuki S, Rosenthal JT, Shaw BW, Atchison RW, et al. Reversibility of lymphomas and lymphoproliferative lesions developing under cyclosporin-steroid therapy. Lancet. 1984; 323 (8377):583-587. doi: 10.1016/S0140-6736(84)90994-2
- Klein G, Purtilo D. Summary: symposium on Epstein-Barr virus-induced lymphoproliferative diseases in immunodeficient patients. Cancer Res. 1981; 41(11 Pt 1): 4302-4304.
- Cockfield SM. Identifying the patient at risk for post-transplant lymphoproliferative disorder. Transplant Infect Dis. 2001; 3: 70-78. doi: 10.1034/j.1399-3062.2001.003002070.x
- Opelz G, Döhler B. Lymphomas after solid organ transplantation: a collaborative transplant study report. Am J Transplant. 2004; 4(2): 222-230. doi: 10.1046/j.1600-6143.2003.00325.x
- Reshef R, Luskin MR, Kamoun M, Heitjan DF, Hadjiliadis D, Goral S, et al. Association of HLA polymorphisms with post-transplant lymphoproliferative disorder in solid-organ transplant recipients. Am J Transplant. 2011;11(4):817-825. doi: 10.1111/j.1600-6143.2011.03454.x
- Pourfarziani V, Einollahi B, Taheri S, Nemati E, Nafar M, Kalantar E, et al. Associations of Human Leukocyte Antigen (HLA) haplotypes with risk of developing lymphoproliferative disorders after renal transplantation. Ann Transplant. 2007;12(4):16-22. PMID: 18344933
- Subklewe M, Marquis R, Choquet S, Leblond V, Garnier JL, Hetzer R, et al. Association of human leukocyte antigen haplotypes with posttransplant lymphoproliferative disease after solid organ transplantation. Transplantation. 2006;82(8):1093-1100.
doi: 10.1097/01.tp.0000235889.05171.12 - Al-Mansour Z, Nelson BP, Evens A.M. Post-transplant lymphoproliferative disease (PTLD): Risk factors, diagnosis, and current treatment strategies. Curr Hematol Malig Rep. 2013; 8(3):173-183. doi: 10.1007/s11899-013-0162-5
- Jagadeesh D, Woda BA, Draper J, Evens A.M. Posttransplant lymphoproliferative disorders: Risk, classification, and therapeutic recommendations. Curr Treat Options Oncol. 2012;13(1):122-136. doi: 10.1007/s11864-011-0177-x
- Ghobrial IM, Habermann TM, Maurer MJ, Geyer SM, Ristow KM, Larson TS et al. Prognostic analysis for survival in adult solid organ transplant recipients with post-transplantation lymphoproliferative disorders. J Clin Oncol. 2005;23(30):7574-7582. doi: 10.1200/JCO.2005.01.0934
- Evens AM, David KA, Helenowski I, Nelson BK, Dixon K, Sheetal M, et al. Multicenter analysis of 80 solid organ transplantation recipients with post-transplantation lymphoproliferative disease: Outcomes and prognostic factors in the modern era. J Clin Oncol. 2010;28(6):1038-1046. doi: 10.1200/JCO.2009.25.4961
- Gavrilina OA, Troitskaya VV, Zvonkov EE, Parovichnikova EN, Galstyan GM, Biryukova LS, et al. EBV-positive central nervous system lymphoproliferative disease associated with immunosuppression after organ transplantation: Long-term remission without chemotherapy. Ter Arkh. 2017;89(7):69-75. (In Russian). doi: 10.17116/terarkh201789769-75
- Reshef R, Vardhanabhuti S, Luskin MR, Heitjan DF, Hadjiliadis D, Goral S, et al. Reduction of immunosuppression as initial therapy for posttransplantation lymphoproliferative disorder. Am J Transplant. 2011;11(2):336-347. doi: 10.1111/j.1600-6143.2010.03387.x
- Zimmermann H, Trappe RU. Therapeutic options in post-transplant lymphoproliferative disorders. Vol. 2, Therapeutic Advances in Hematology. SAGE Publications; 2011. p. 393-407. doi: 10.1177/2040620711412417
- Lister J, Simpson JK, DeMagalhaes-Silverman MM, Rybka WB, Donnenberg AD, Myers DJ, et al. Allogeneic peripheral blood stem cell transplant for myelodysplasia after chemotherapy for post-transplant lymphoma in a cardiac transplant recipient at 10 years. Bone Marrow Transplant. 1997;19(9):943-945. doi: 10.1038/sj.bmt.1700758
- Kim M.J, Kim I, Bae HM, Seo K, Park N, Yoon SS, et al. Hematopoietic stem cell transplantation after posttransplant lymphoproliferative disorder. J Korean Med Sci. 2010;25(5):781-784. doi: 10.3346/jkms.2010.25.5.781
- Schechter-Finkelstein T, Gassas A, Weitzman S, Grant D, Pollock-Barziv S, Dipchand A, et al. Hematopoietic stem-cell transplantation following solid-organ transplantation in children. Bone Marrow Transplant. 2011;46(10):1321-1325. doi: 10.1038/bmt.2011.153
- Gabeeva NG, Zvonkov EE, Koroleva DA, Chukavina MM, Obukhova TN, Kovrigina AM et al. Successful experience of treatment of a patient with generalized non-GCB-DLBCL using the R-mNHL-BFM-90 protocol with lenalidomide: Case report and review of literature. Ter Arkh. 2018;90(7): 96-101. (In Russian). doi: 10.26442/terarkh201890796-101
- Nowakowski GS, LaPlant B, Macon WR, Reeder CB, Foran JM, Nelson GD, et al. Lenalidomide combined with R-CHOP overcomes negative prognostic impact of non-germinal center B-cell phenotype in newly diagnosed diffuse large B-cell lymphoma: A phase II study. J Clin Oncol. 2015;33(3):251-257. doi: 10.1200/JCO.2014.55.5714
Accepted 18 March 2022


