Caspofungin as secondary prophylaxis or therapy in patients undergoing allogeneic stem cell transplantation with a prior or ongoing history of systemic or invasive fungal infections
N. Stute, T. Zabelina, N. Fehse, W. Hassenpflug, J. Panse, C. Wolschke, F. Ayuk, H. Schieder, H. Renges, A. Kratochwille, R. von Hinüber, R. Erttmann, A.R. Zander, N. Kröger
Dept of Stem Cell Transplantation, University Hospital Hamburg-Eppendorf, 20246 Hamburg, Germany
Correspondence:
Prof. Dr. med. Nicolaus Kröger, Bone Marrow Transplant Center, University Hospital Hamburg-Eppendorf, Martinistr. 52, D-20246 Hamburg, Germany, Tel.: +49-40-42803 5864, Fax: +49-40-42803 3795, E-mail: nkroeger@uke.uni-hamburg.de
Accepted 21 November 2008
Published 28 November 2008
Summary
Background
Patients with a history of or ongoing invasive fungal infection (IFI) who undergo allogeneic stem cell transplantation (SCT) have a high risk of reactivation or progression. In a prospective study we evaluated the efficacy and safety of caspofungin as secondary prophylaxis or as therapy for persistent disease.
Methods
Twenty-eight adult patients were included in this study, all of whom had acute leukemia. At the time of SCT 16 patients had no signs of infection, while in 12 cases radiographic signs (CT scan) of florid fungal infections were noted. Caspofungin 50 mg intravenously was given daily from start of conditioning until stable engraftment.
Results
No patient experienced side effects leading to the discontinuation of caspofungin. In 14 out of 16 patients (88%) without active signs of infection at start of transplantation, no fungal disease was observed after prophylaxis with caspofungin. In 10 out of 12 cases (83%) with radiographic signs of florid fungal infection pre-transplantation, complete (n=4) or partial (n=6) responses after caspofungin treatment were achieved.
Conclusions
The use of caspofungin is safe and effective in high-risk patients with a history of IFI when undergoing allogeneic SCT.
Keywords
Prophylaxis, invasive fungal infection (ifi), infectious complications, caspofungin, allogeneic stem cell transplantation
Introduction
Fungal infections with Aspergillus or Candida, mainly of the lung, are a major cause of treatment-related mortality in allogeneic stem cell transplantation (SCT) [1, 2]. Invasive Aspergillosis (IA) in hematopoietic stem cell transplant (HSCT) recipients was associated with case-fatality rates of 87% [3], and an overall 1-year survival rate of only 20% [4]. Patients with a history of systemic or invasive fungal infection (IFI) who undergo allogeneic transplantation are considered to be at high risk of disease reactivation or progression leading to a high mortality. More recently, leukemias and other malignancies have been treated with more intensive induction therapies up front, resulting in a higher incidence of fungal infections before receiving a stem cell transplant.
Primary antifungal prophylaxis is given routinely in patients with no prior IFI who undergo allogeneic stem cell transplantation [5-9], whereas secondary prophylaxis is given in patients with prior IFI. Up to now, for those patients with a history of IFI who undergo allogeneic SCT, no prospective study of secondary antifungal prophylaxis in a larger series of these high-risk patients exists. More recently, several retrospective studies indicate that bone marrow transplantation (BMT) in patients with a history of IA is feasible [10-12]. Apart from that, the only case reports that can be found in the literature concern patients with fungal disease who underwent SCT and were successfully treated with fluconazole, itraconazole, surgical resection, amphotericin B, and voriconazole [13-15].
We therefore conducted a prospective single-center study to evaluate the efficacy and safety of caspofungin as secondary prophylaxis or therapy, in order to prevent recurrence or progression of systemic or invasive fungal infections in patients with a history of IFI who underwent allogeneic SCT.
Caspofungin is an echinocandin, which interferes with fungal cell wall assembly by inhibition of β(1,3)-D-glucan synthase, an enzyme not present in mammalian cells [16]. It shows no signs of nephrotoxicity and has only modest side effects, such as chills and fever, in very few patients (1-3 %). Caspofungin has proven to be effective and well-tolerated in the treatment of infections caused by Aspergillus and Candida species in clinical trials [17-24].
Patients and methods
Study design
Patients included in our study had a history of proven or probable IFI according to modified EORTC criteria for diagnosis of invasive fungal infection. Proven IFI requires histo- or cytopathology positive for hyphae with evidence of associated tissue damage or a positive culture for mold species from a normally sterile site consistent with infection. Probable IFI included cases with one host criterion plus one microbial criterion and one major or two minor clinical criteria, or a patient with recent neutropenia or an allogeneic SCT patient with a chest CT scan positive for "halo" or "air crescent" signs [25-28]. The patients included were scheduled to undergo an allogeneic bone marrow or stem cell transplantation and needed to have sufficient organ function.
The primary efficacy endpoint was the incidence of clinically manifest mycoses under the prophylactic use of caspofungin (development of breakthrough IFI) or response thereof, in case of florid infections at start of conditioning. The secondary endpoints were the evaluation of toxicity and overall survival until day +365. Failure was defined as suspected or documented fungal infection (failure to prevent a new IFI), or a progressive fungal infection in case of prior florid activity (failure to improve a partially treated IFI). Secondary endpoints were evaluated descriptively. All patients included were evaluated on an intent-to-treat basis.
Modified EORTC criteria for diagnosis of IFI are more clinically oriented than the strict EORTC-IFICG/MSG criteria (European Organization for Research and Treatment of Cancer - IFI Cooperative Group & Mycoses Study Group). These criteria were specifically developed for use in large clinical trials on the efficacy of antifungals and should not be used to guide clinical decisions [25]. This approach is reflected by the clinical practice in BMT, where proven and even probable IFI with direct microbiological evidence are diagnosed in only a minority of high risk patients, and diagnosis of fungal infection is often made on clinical and radiological grounds alone.
Prior to study entry, we performed a chest CT scan, a bronchoalveolar lavage (BAL), and an abdominal ultrasound. We also carried out a high-resolution chest CT scan at the end of the caspofungin prophylaxis or in case of fever. Weekly controls of circulating Aspergillus galactomannan antigen (Platelia® Aspergillus EIA, index used for cut-off was 1.0, BioRad Laboratories, Hercules, CA, USA) and Candida antibody titer (Cand-Tec® latex agglutination test for heat-labile antigen, Ramco Laboratories, Stafford, TX, USA) were performed in all patients. To measure potential side effects we conducted daily blood counts and clinical chemistry analyses.
Patients
Successive patients with a history of systemic or invasive fungal infection in the past who underwent allogeneic BMT were included in this prospective study.
Conditioning was performed with myeloablative regimens. In unrelated donors and mismatched family donors, we added anti-thymocyte globulin (ATG Fresenius, 30-90 mg/kg intravenously (i.v.)) to the conditioning in order to minimize the risk of graft-versus-host disease (GvHD). Cyclosporin A (CSA) for immunosuppression was given to all patients post-transplant with a target level of 200-300 µg/L. The local ethics committee approved the study and all patients gave prior written informed consent.
Treatment plan & response assessment
Patients were given 50 mg caspofungin i.v. daily over one hour, following a 70 mg loading dose from start of conditioning until stable engraftment. No other systemic antifungal was given. All patients were nursed in reversed isolation in conventional or laminar airflow rooms. Antibiotic prophylaxis consisted of ciprofloxacin, local antifungals, and acyclovir. Prophylaxis against pneumocystis carinii was carried out with cotrimoxazole (or pentamidine inhalation). CMV-negative patients received only CMV-negative blood products. All blood products were irradiated at 25 Gy. CMV-positive patients were monitored weekly for CMV infection by PCR and/or antigenemia-assay for pp65. Preemptive therapy was started with 10 mg/kg gancyclovir after two consecutive positive PCR or one positive antigenemia-assay. In the case of neutropenic fever during transplantation, ceftazidime and vancomycin were given; if fever or signs of infection persisted, tobramycin was added after two days. In patients without signs of fungal infection after engraftment, antimycotic prophylaxis was switched to itraconazole 200 mg twice daily until day +100.An experienced clinician and radiologist performed the mycological response assessment according to EORTC criteria. Radiographic criteria for complete response (CR) required a > 90% clearing of CT scan abnormalities associated with active fungal infection or persistent residual scarring only. Partial response (PR) required a > 50% improvement in radiographic abnormalities on relevant scans, as compared to baseline. Stable response was diagnosed if minor or no improvement occurred and there was no worsening of attributable radiographic signs of the IFI. Failure included worsening CT scan abnormalities consistent with progressive infection or, clinically, an increase in the number and/or severity of clinical signs and symptoms attributable to the IFI.
Results
Patients' characteristics
Twenty-eight successive patients from one center, all of whom happened to have acute leukemias, were included in this prospective study from July 2001 to September 2003 (details of patients are given in Table 1). Patients included in this study had a history of probable (n=26) or proven (n=2) systemic or invasive fungal infection in the past according to modified EORTC criteria, which include typical signs of fungal infection on a CT scan closely related to neutropenia.
Prior fungal infection was diagnosed by a CT scan of the lungs (n=27) and liver, spleen or kidneys (n=9) at a median of 3 months before transplant (range 1 week – 3 years).
Table 1. Patients' characteristics
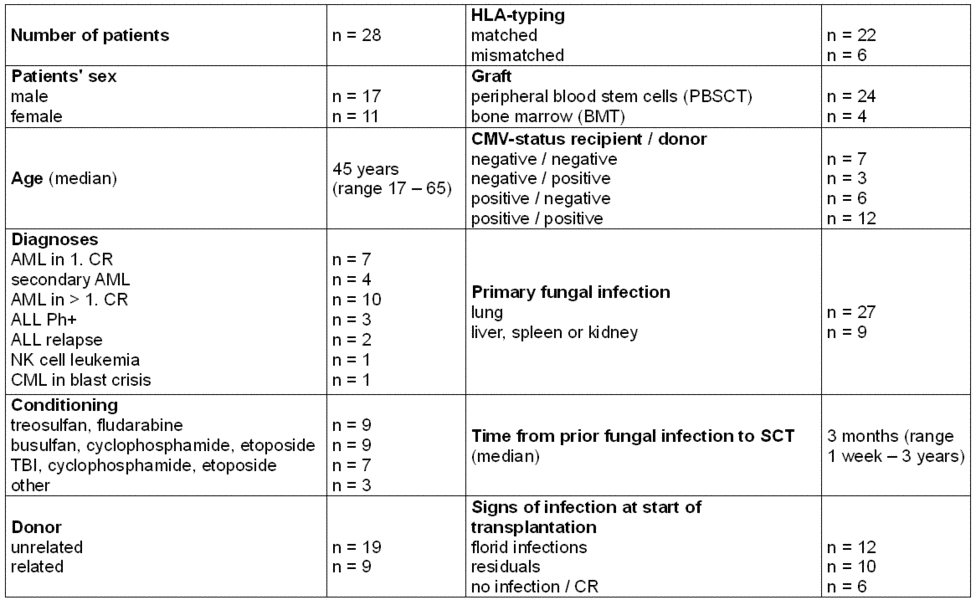
At the beginning of transplantation, 12 patients (often unexpectedly) had florid infections by CT scan criteria, 10 had residuals and 6 were in CR. A bronchoscopy (BAL) and an abdominal ultrasound were performed prior to conditioning in most patients, but were not as informative as high resolution CT scans. However, in two patients with residual disease, we isolated Candida albicans via BAL in one, and Candida glabrata in the other.
Conditioning was performed with myeloablative regimens. Median duration of neutropenia was 18 days (range 7-47). Transplants were from unrelated (n=19) and related (n=9) donors, 6 of whom had a mismatch.
Side effects and toxicity of caspofungin
Caspofungin was given over a median duration of 27 days (range 15-47), which was similar in patients with and without active signs of IFI at the start of caspofungin. It was not necessary to discontinue caspofungin due to side effects in any patient. Median toxicity of conditioning, immunosuppression and supportive therapy according to Bearman was comparable to historic controls: mouth: grade 2, liver: grade 1, kidney: grade 0, and gut: grade 0. Maximal laboratory values (median and range) for liver and renal toxicity during caspofungin prophylaxis or therapy were: total bilirubin 2.7 mg/dL (range 1.2-22.2, normal ≤ 1.0), AST 36 U/L (range 6-215, normal ≤ 18), ALT 46 U/L (range 9-299, normal ≤ 22), and creatinine 1.1 mg/dL (range 0.7-3.0, normal ≤ 1.2). When maximal bilirubin was > 6 (n=4) mg/dL it was either due to liver toxicity (n=3), not related to any particular drug, or suspected hepatic veno-occlusive disease (n=1). When maximal creatinine was > 2 mg/dL (n=6) it was usually due to the nephrotoxic side effects of antibiotics given for fever and CSA. It is noteworthy that caspofungin was well tolerated in all 28 patients who received CSA for immunosuppression post-transplant without any associated renal or hepatic problems. No CSA dosage adaptation was required due to liver toxicity potentially related to caspofungin. Ten out of 28 patients experienced acute GvHD ≥ grade 2 after engraftment and prior to day +100 and needed additional immunosuppression.
Response to caspofungin
Importantly, in 10 out of 12 cases (83%) a florid fungal infection with a positive CT scan at start of transplantation responded to caspofungin alone, with 4 CR and 6 PR despite severe immunosuppression (Table 2).
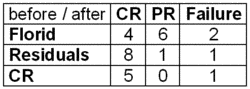
Table 2. Response to caspofungin after engraftment by CT scan criteria.
NOTE. CR = complete response, PR = partial response (for definition see Methods)
Two case reports are shown for illustration (Figure 1). Four out of 12 patients received additional granulocyte transfusions during aplasia, and one patient with prolonged aplasia of 29 days nevertheless developed a fungal infection. In 14 out of 16 patients (88 %) without active signs of infection at start of transplantation, no fungal disease was observed after prophylaxis with caspofungin. Only 2 out of 16 patients developed a fungal infection under caspofungin despite additional granulocyte transfusions. Both patients had prolonged aplasia for 40 and 47 days, respectively.
Figure 1. Case reports of responses to caspofungin. Shown are chest CT scans of patients undergoing stem cell transplantation prior conditioning and after engraftment.
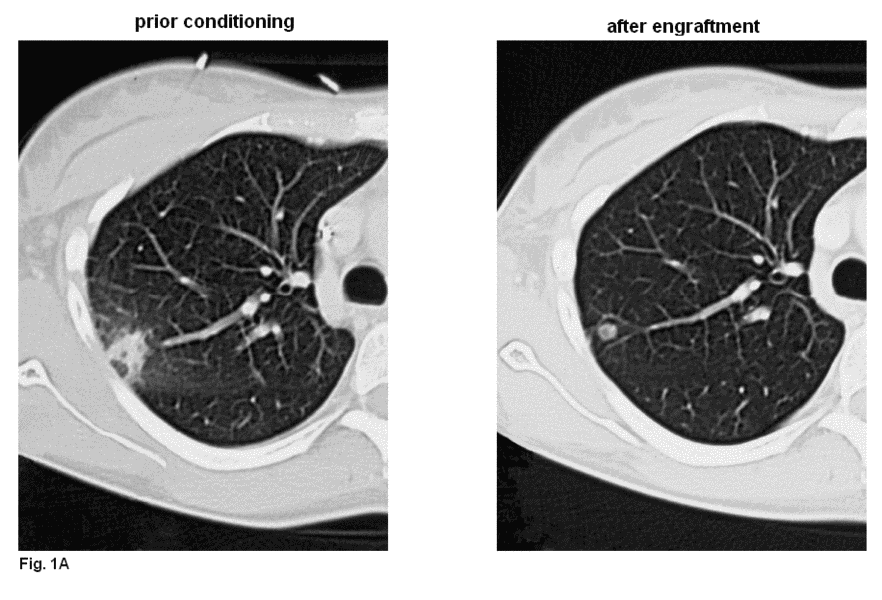
A Partial response of prior florid fungal infection: 29-year-old woman with AML, FAB M6, complex cytogenetics. She received a mismatched unrelated PBSCT and had 23 days of aplasia.
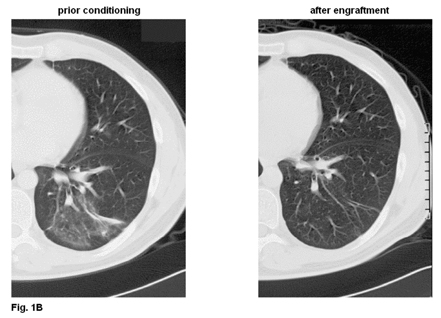
B Complete response of prior florid fungal infection: 26-year-old man with early relapse c-ALL. He was given a matched unrelated BMT and had 22 days of aplasia.
Treatment failure was defined as documented or suspected progressive fungal infection.
In 4 out of 28 patients (1 CR, 1 residual and 2 florid states prior to transplantation) anti-mycotic treatment was, therefore, changed from caspofungin to second-line agents such as voriconazole, amphotericin B or AmBisome® i.v. (= 14% failure rate). Three out of four patients who developed a fungal infection following HSCT despite prophylaxis or therapy with caspofungin had delayed engraftment or primary graft failure with 29, 40 and 47 days of aplasia with leucocytes <1000 /µl.
The four patients who failed on caspofungin had suspected or documented progressive fungal infection until engraftment. All four had a positive chest CT scan with worsening of radiographic signs and only one had positive antigen tests for IFI. Patient 1 had increasing CRP, a positive test for Aspergillus antigen, and 40 days of aplasia due to primary graft failure. She received granulocytes and her autologous backup, responded to voriconazole and amphotericin B (PR), but still had residuals at day +100 and developed new infiltrates later. Patient 2 was BAL negative. She responded to voriconazole initially (PR) but died before day +100 from alveolar hemorrhage with pancytopenia and suspected fungal pneumonia. Patient 3 had a fever, Aspergillus fumigatus in BAL, and a normal antifungal serology (failure of secondary prophylaxis with fungal breakthrough infection). He had 47 days of aplasia, received granulocytes, and had grade 2 toxicity of mouth, liver, and kidney. He was treated with amphotericin B and had a CR from his fungal disease by day +100. However, he later died from a relapse of his leukemia. Patient 4 had 29 days of aplasia. She was given AmBisome®, responded with a CR, but died of multi-organ failure prior to day +100.
One patient with florid nodular infiltrates responded to caspofungin initially (PR), but received high dose steroids because of severe acute skin GvHD after engraftment, and later died of systemic Scedosporium prolificans resistant to caspofungin.
1 Year outcome
Overall mortality at one year was as high as expected. 11 out of 28 patients (39%) had died with overall survival of 61%. Causes of death were: pneumonia with sepsis (n=3) with suspected fungal pneumonia in two cases, systemic fungal sepsis with Scedosporium (n=1), sepsis of unknown etiology (n=1), alveolar hemorrhage (n=1), multi-organ failure (n=2), and relapse of leukemia (n=3). Five deaths were associated with pancytopenia or secondary graft failure and five fatalities with severe GvHD. Transplant-related mortality (TRM) was 8 out of 28 patients (29%) and no death was attributed to caspofungin. Six out of 8 patients who died due to TRM within the first year had signs of pulmonary infiltrates at time of death, and in 2 cases these were indicative of active IFI (patients A and B, see below). Only 1 out of 17 patients still alive at 1 year had signs of residual fungal disease (patient C, see below).
Relapse IFI of the lung in the first year post transplant after the initial phase of caspofungin prophylaxis or therapy was suspected in 3 patients (patients who had a relapse of their leukemia prior to developing signs of IFI are not included): At 1.5 months after transplant, patient A, after being treated with steroids for skin and gut GvHD, still under itraconazole prophylaxis, developed atypical infiltrates on chest CT scan. He was treated with antibiotics and voriconazole, and died of pneumonia shortly thereafter. Eight months after transplant, patient B, after a cholecystitis and colitis, showed a positive chest X-ray and voriconazole treatment was initiated; he developed an ileus and died shortly thereafter of multi-organ failure. After 8 and 10 months, patient C (caspofungin-failure patient 1), after a relapse of her leukemia and another SCT with primary graft failure, developed new round infiltrates on chest CT scan, which regressed under therapy with voriconazole.
Six out of 28 patients (21%) experienced a relapse of their leukemia in the first year after transplant. Three patients underwent a second SCT and achieved a complete remission of their leukemia. One patient progressed despite reinduction therapy and two patients received palliative therapy only. At one year, all 17 patients alive were in complete remission with regard to their underlying disease.
Discussion
This is the first prospective, non-controlled, single-center study of secondary antifungal prophylaxis or therapy in allogeneic SCT in high-risk patients with prior systemic or invasive fungal infections. Caspofungin is effective for secondary prophylaxis and in those patients with an active infection at time of SCT. In 88% of patients (n=16) without active signs of infection at start of transplantation, secondary prophylaxis with caspofungin was successful, and in 83% of patients (n=12), a florid fungal infection at the time of transplantation responded to caspofungin despite aplasia and additional immunosuppression. Scheduled transplantations were not delayed until fungal infections resolved, for this may have had major implications for prognosis.
Six patients received 1 to 6 granulocyte transfusions (median 3) before engraftment, which could also attribute to the resolution of fungal infection. Three patients failed on caspofungin despite receiving granulocytes. Each of these patients had prolonged aplasia >4 weeks (one had signs of florid infection, one had residuals, and one was in CR at the start of transplantation).
Retrospective studies had indicated that BMT in patients with a history of IA is feasible. Offner et al. performed a multicenter retrospective analysis of 48 patients with documented or probable IA prior to BMT using various antifungals for secondary prophylaxis. The overall incidence of relapse IA was lower than expected (33%), but the mortality rate among relapsed patients was 88% [10]. Fukuda et al. described a 10-year experience at a single transplant center and included 45 patients with a known history of IA before HSCT. Post-transplantation IA occurred in 13 of these patients, 9 infections were considered recurrent. Compared with other patients who received allogeneic HSCT during the same period, patients with histories of IA had a lower overall survival of 56% and a higher TRM of 38% at 100 days after BMT [11].
Post-transplantation IA occurred more frequently in patients who received < 1 month of antifungal therapy prior to BMT. Patients receiving >1 month of antifungal therapy and who had a resolution of radiographic abnormalities did better. Recently, Cordonnier et al. have shown that voriconazole may be useful to prevent reactivation of prior fungal disease in a retrospective study of 9 patients undergoing allogeneic HSCT [12]. These included 5 proven and 4 probable cases of IA according to EORTC criteria and at time of transplant, 3 patients had residual fungal disease. Voriconazole 400 mg/day i.v. or p.o. was given from start of conditioning until end of immunosuppression. None of the patients experienced fungal relapse or new fungal disease and scheduled treatment was not delayed. Voriconazole was well tolerated, except in one patient who had abnormal liver tests secondary to GvHD, and one who had transient visual disturbances.
Caspofungin was chosen in our prospective study as secondary prophylaxis or therapy because of its high efficacy in Aspergillus and Candida infections and its low side effects [22]. Caspofungin is active in vitro against Candida spp., including C. krusei, C. glabrata and C. tropicalis, as well as Aspergillus spp., such as A. fumigatus, A. flavus and A. niger. Caspofungin is resistant, however, against Fusarium, Rhizopus, Cryptococcus and Scedosporium prolificans [22]. Caspofungin has shown high efficacy in clinical trials in esophageal candidiasis, refractory invasive aspergillosis, candidemia, and empiric antifungal therapy. It has been approved for first-line therapy of candidemia and second-line therapy of aspergillosis [17-23].
Sable et al. analyzed the safety and tolerability of caspofungin, which was generally well tolerated; caspofungin was discontinued only in 2% of patients due to adverse events [24]. Drug-related adverse events were fever, local phlebitis at the infusion-site, headache and nausea. Renal tolerability was excellent and transient mild-to-moderate elevations in ALT, AST and alkaline phosphatase levels were observed. In our study, caspofungin was well tolerated and did not have to be discontinued due to side effects in a single patient. It is of note that caspofungin was safe in all 28 patients who received cyclosporin A (CSA) for immunosuppression post-transplant without any liver or renal problems due to CSA. Similar results have been found by Sanz-Rodriguez et al. recently in 13 patients who received CSA concomitantly [29]. The incidence of liver and renal toxicity and acute GvHD ≥ grade 2 was comparable to historic controls without caspofungin.
Long-term aplasia is a high risk factor for fungal (breakthrough) infection. Chest CT scans prior to transplantation and frequently thereafter, e.g. once weekly, are likely to benefit this high-risk group of patients. In our study, in 12 out of 28 cases chest CT scans revealed a florid infection at start of transplantation despite a lack of clinical signs and symptoms.
A limitation of our study was that no control group existed; therefore conclusions about toxicity and efficacy are based on clinical experience and should be handled with caution. There also was a tendency to declare failure in neutropenic patients who had increasing infiltrates on CT scan, and to discontinue caspofungin early despite the lack of concrete evidence of IFI (this was possibly relevant in 3 out of 4 patients).
In conclusion, the use of caspofungin as secondary prophylaxis or therapy in high-risk patients with a history or persistence of IFI undergoing allogeneic SCT is safe and effective. It does not result in a high toxicity and shows a low incidence of breakthrough infections. This allows SCT to proceed as scheduled, even if signs of florid fungal disease are still evident at time of transplant.
Acknowledgements
We thank the staff of the BMT unit for providing excellent care of our patients, the radiologists for their cooperation, and the medical technicians for their excellent work in the BMT laboratory.
References
1. Fukuda T, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG, et al. Risks and outcomes of invasive fungal infections in recipients of allogeneic hematopoietic stem cell transplants after nonmyeloablative conditioning. Blood. 2003;102(3):827-833.
2. Wingard JR. Antifungal chemoprophylaxis after blood and marrow transplantation. Clin Infect Dis. 2002;34(10):1386-1390.
3. Lin SJ, Schranz J, Teutsch SM. Aspergillosis case-fatality rate: systematic review of the literature. Clin Infect Dis. 2001;32(3):358-366.
4. Marr KA, Carter RA, Crippa F, Wald A, Corey L. Epidemiology and outcome of mould infections in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2002;34(7):909-917.
5. Hamza NS, Ghannoum MA, Lazarus HM. Choices aplenty: antifungal prophylaxis in hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2004;34(5):377-389.
6. Goodman JL, Winston DJ, Greenfield RA, Chandrasekar PH, Fox B, Kaizer H, et al. A controlled trial of fluconazole to prevent fungal infections in patients undergoing bone marrow transplantation. N Engl J Med. 1992;326(13):845-851.
7. Slavin MA, Osborne B, Adams R, Levenstein MJ, Schoch HG, Feldman AR, et al. Efficacy and safety of fluconazole prophylaxis for fungal infections after marrow transplantation - a prospective, randomized, double-blind study. J Infect Dis. 1995;171(6):1545-1552.
8. Marr KA, Seidel K, Slavin MA, Bowden RA, Schoch HG, Flowers ME, et al. Prolonged fluconazole prophylaxis is associated with persistent protection against candidiasis-related death in allogeneic marrow transplant recipients: long-term follow-up of a randomized, placebo-controlled trial. Blood. 2000;96(6):2055-2061.
9. Dykewicz CA. Summary of the Guidelines for Preventing Opportunistic Infections among Hematopoietic Stem Cell Transplant Recipients. Clin Infect Dis. 2001;33(2):139-144.
10. Offner F, Cordonnier C, Ljungman P, Prentice HG, Engelhard D, De Bacquer D, et al. Impact of previous aspergillosis on the outcome of bone marrow transplantation. Clin Infect Dis. 1998;26(5):1098-1103.
11. Fukuda T, Boeckh M, Guthrie KA, Mattson DK, Owens S, Wald A, et al. Invasive aspergillosis before allogeneic hematopoietic stem cell transplantation: 10-year experience at a single transplant center. Biol Blood Marrow Transplant. 2004;10(7):494-503.
12. Cordonnier C, Maury S, Pautas C, Bastie JN, Chehata S, Castaigne S, et al. Secondary antifungal prophylaxis with voriconazole to adhere to scheduled treatment in leukemic patients and stem cell transplant recipients. Bone Marrow Transplant. 2004;33(9):943-948.
13. Martino R, Nomdedeu J, Altes A, Sureda A, Brunet S, Martinez C, et al. Successful bone marrow transplantation in patients with previous invasive fungal infections: report of four cases. Bone Marrow Transplant. 1994;13(3):265-269.
14. Richard C, Romon I, Baro J, Insunza A, Loyola I, Zurbano F, et al. Invasive pulmonary aspergillosis prior to BMT in acute leukemia patients does not predict a poor outcome. Bone Marrow Transplant. 1993;12(3):237-241.
15. Mattei D, Mordini N, Lo NC, Ghirardo D, Ferrua MT, Osenda M, et al. Voriconazole in the management of invasive aspergillosis in two patients with acute myeloid leukemia undergoing stem cell transplantation. Bone Marrow Transplant. 2002;30(12):967-970.
16. Denning DW. Echinocandin antifungal drugs. Lancet. 2003;362(9390):1142-1151.
17. Villanueva A, Arathoon EG, Gotuzzo E, Berman RS, DiNubile MJ, Sable CA. A randomized double-blind study of caspofungin versus amphotericin for the treatment of candidal esophagitis. Clin Infect Dis. 2001;33(9):1529-1535.
18. Villanueva A, Gotuzzo E, Arathoon EG, Noriega LM, Kartsonis NA, Lupinacci RJ, et al. A randomized double-blind study of caspofungin versus fluconazole for the treatment of esophageal candidiasis. Am J Med. 2002;113(4):294-299.
19. Arathoon EG, Gotuzzo E, Noriega LM, Berman RS, DiNubile MJ, Sable CA. Randomized, double-blind, multicenter study of caspofungin versus amphotericin B for treatment of oropharyngeal and esophageal candidiases. Antimicrob Agents Chemother. 2002;46(2):451-457.
20. Mora-Duarte J, Betts R, Rotstein C, Colombo AL, Thompson-Moya L, Smietana J, et al. Comparison of caspofungin and amphotericin B for invasive candidiasis. N Engl J Med. 2002;347(25):2020-2029.
21. Maertens J, Boogaerts M. Caspofungin in the treatment of candidosis and aspergillosis. Int J Infect Dis. 2003;7(2):94-101.
22. Letscher-Bru V, Herbrecht R. Caspofungin: the first representative of a new antifungal class. J Antimicrob Chemother. 2003;51(3):513-521.
23. Walsh TJ, Teppler H, Donowitz GR, Maertens JA, Baden LR, Dmoszynska A, et al. Caspofungin versus liposomal amphotericin B for empirical antifungal therapy in patients with persistent fever and neutropenia. N Engl J Med. 2004;351(14):1391-1402.
24. Sable CA, Nguyen BY, Chodakewitz JA, DiNubile MJ. Safety and tolerability of caspofungin acetate in the treatment of fungal infections. Transpl Infect Dis. 2002;4(1):25-30.
25. Ascioglu S, Rex JH, de Pauw B, Bennett JE, Bille J, Crokaert F, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis. 2002;34(1):7-14.
26. Caillot D, Couaillier JF, Bernard A, Casasnovas O, Denning DW, Mannone L, et al. Increasing volume and changing characteristics of invasive pulmonary aspergillosis on sequential thoracic computed tomography scans in patients with neutropenia. J Clin Oncol. 2001;19(1):253-259.
27. Kuhlman JE, Fishman EK, Burch PA, Karp JE, Zerhouni EA, Siegelman SS. Invasive pulmonary aspergillosis in acute leukemia. The contribution of CT to early diagnosis and aggressive management. Chest. 1987;92(1):95-99.
28. Kuhlman JE, Fishman EK, Siegelman SS. Invasive pulmonary aspergillosis in acute leukemia: characteristic findings on CT, the CT halo sign, and the role of CT in early diagnosis. Radiology. 1985;157(3):611-614.
29. Sanz-Rodriguez C, Lopez-Duarte M, Jurado M, Lopez J, Arranz R, Cisneros JM, et al. Safety of the concomitant use of caspofungin and cyclosporin A in patients with invasive fungal infections. Bone Marrow Transplant. 2004.
Accepted 21 November 2008
Published 28 November 2008


